Select a Regulatory Authority (for countries where there is no national regulatory authority, ADD icon is not available)
Regulatory Authority Name
If Regulatory Authority Name is missing in Regulatories Authorities, you have (if you have the right) to add the name at the country level in REGULATORIES option.
If you want to reset the entered information for a document, click on
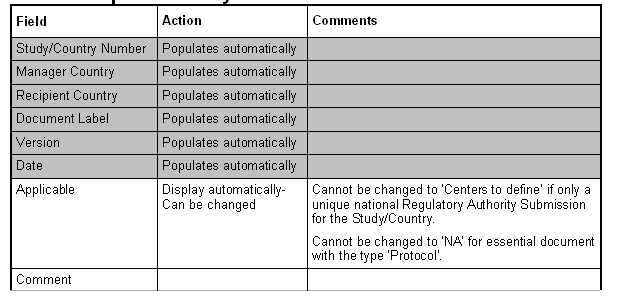
List of Essential Documents
Enter Regulatory Authority
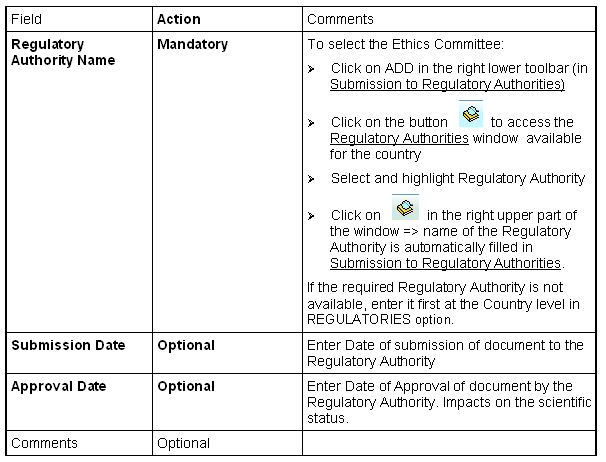
If Regulatory Authority Submission type is multiple for the national level, the ADD button in the right lower horizontal toolbar becomes active as soon as Regulatory Authority name has been entered for the first document; thus you can 'submit' the document to several Regulatories Authorities.
Option in Submission to Regulatory Authorities: